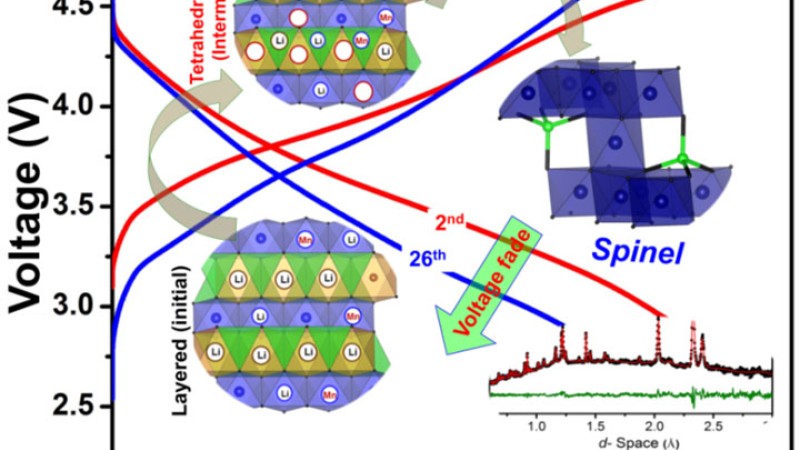
Neutron diffraction tracked the cation migration paths that trigger a structural transformation responsible for voltage fade in high-voltage Li-Mn-rich (LMR) oxide cathodes.
The layered-to-spinel structural transformation in LMR oxides degrades the voltage profile with electrochemical cycling, thereby limiting its energy-density. By understanding how this “atomic restructuring” occurs in LMR oxides, it is possible to prevent it by manipulating the composition/structure. This could open the door for practical use of these high-energy-density lithium-ion batteries.
LMR cathode samples were obtained from pouch cells after electrochemical cycling into different charge states. Neutron diffraction refinement provided the cation occupancies (with exceptional sensitivity to Lithium ions) at different charge states. The transformation occurs via i) intralayer Li migration from octahedral to tetrahedral sites and ii) interlayer Mn migration from octahedral sites in the transition metal layer to octahedral sites in the lithium layer via tetrahedral sites.
Work was performed at the ORNL Spallation Neutron Source’s POWGEN instrument, which is a DOE Office of Science User Facility. This material is based upon work supported by the U.S. Department of Energy, Office of Energy Efficiency and Renewable Energy, Office of Vehicle Technologies ‘s Applied Battery Research Program.
D. Mohanty, J. Li, D.P. Abraham, A. Huq, E. Andrew Payzant, D. L. Wood III, and C. Daniel. “Unraveling the Voltage-Fade Mechanism in High-Energy-Density Lithium-Ion Batteries: Origin of the Tetrahedral Cations for Spinel Conversion.” Chemistry of Materials. 26, 21 (2014): 6272-6280. http://pubs.acs.org/doi/abs/10.1021/cm5031415